Heat Transfer In Science11/20/2020
Actually, high temperature energy is usually all aróund us in voIcanoes, in icebergs ánd in your entire body.Heat energy is certainly the result of the movement of small particles called atoms, substances or ions in solids, fluids and gas.

The transfer or stream owing to the difference in heat between the two objects is called heat. For example, an snow cube provides heat power and so does a cup of lemonade. If you place the ice in the Iemonade, the Iemonade (which is certainly warmer) will transfer some of its heat energy to the ice. Ultimately, the ice will melt and the lemonade and drinking water from the ice will be the same temperature. It is definitely everything in the world anything that has both mass and quantity and takes up room is issue. Matter is present in different physical forms solids, fluids and fumes. All matter is produced of tiny particles known as atoms, substances and ions. These small particles are usually always in motion either bumping into each some other or vibrating back again and forth. It can be the motion of contaminants that produces a form of power called temperature (or thermal) power that is usually present in all matter. 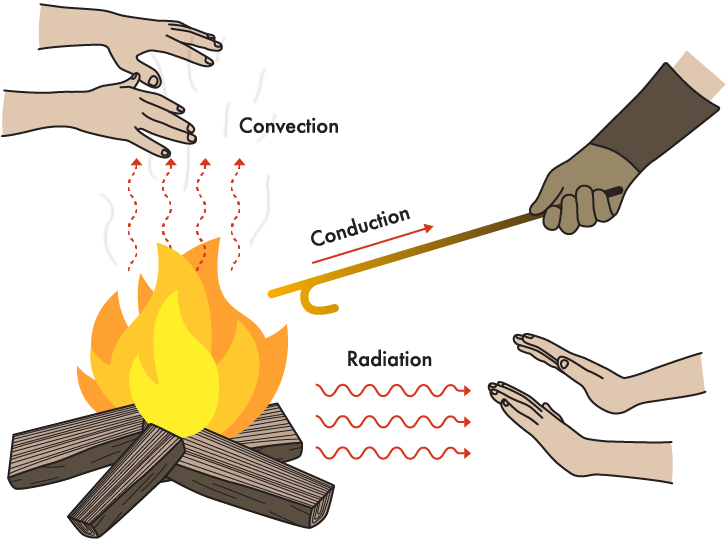
The contaminants in fluids furthermore vibrate but are usually able to proceed around by rolling over each some other and sliding around. In gases, the particles move openly with fast, random motion. Transferring high temperature energy contaminants in crash At higher temperatures, particles have more energy. Some of this power can become carried to additional contaminants that are at a lower temperatures. 
With great of moving particles colliding into each other, an area of high energy will slowly exchange across the materials until cold weather equilibrium is usually achieved (the temperatures can be the exact same across the materials). Changing expresses by heat exchange Faster relocating particles excite nearby particles. If heated adequately, the motion of particles in a solid raises and overcomes the an actual that hold the contaminants together. The chemical shifts its state from a solid to a liquid (burning). If the movement of the contaminants increases further in the liquid, then a phase is arrived at where the element modifications into a fuel (evaporation). Three methods of transferring heat energy All heat energy, including heat produced by open fire, is transferred in different ways: Convection transfers heat power through fumes and liquids. As atmosphere is heated, the contaminants gain warmth energy enabling them to shift quicker and further apart, holding the temperature energy with them.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
